How International Liposomal Innovations Drive WBCIL’s Research Forward?
In the dynamic and ever-evolving landscape of healthcare, pharmaceuticals, nutraceuticals, and cosmeceuticals are increasingly challenged to deliver more effective, targeted, and consumer-friendly solutions. One technology has quietly transformed from a laboratory curiosity into a powerhouse of innovation — liposomal delivery systems. From enhancing nutrient absorption to enabling precision drug targeting, liposomes have revolutionized the way active compounds are delivered into the human body.
Born from the pioneering research of the 1960s, liposomes were originally studied as simple phospholipid bilayer structures mimicking biological membranes. Over the past six decades, this seemingly modest innovation has evolved into a globally recognized platform for next-generation therapies. Today, liposomal technology plays a pivotal role in areas ranging from cancer therapeutics and vaccine delivery to skincare, immune support, and chronic disease management.
What sets liposomes apart is their ability to protect delicate molecules, navigate biological barriers, and ensure higher bioavailability with minimal side effects. This unique advantage has driven global scientific interest, resulting in thousands of publications, patents, and commercial products emerging worldwide.
At West Bengal Chemical Industries Limited (WBCIL), we recognize the transformative potential of liposomes not only in improving the efficacy of pharmaceuticals and nutraceuticals but also in shaping the future of personalized and precision healthcare. Our research and development teams are deeply connected with the pulse of international liposomal innovation, integrating global insights to create high-performance formulations for a rapidly changing market.
In this article, we explore how global advancements in liposomal technology — through scientific research, clinical trials, and international conferences — continuously inspire and shape WBCIL’s innovation journey.

The Evolution of Liposomal Innovation: A Timeline of Scientific Momentum
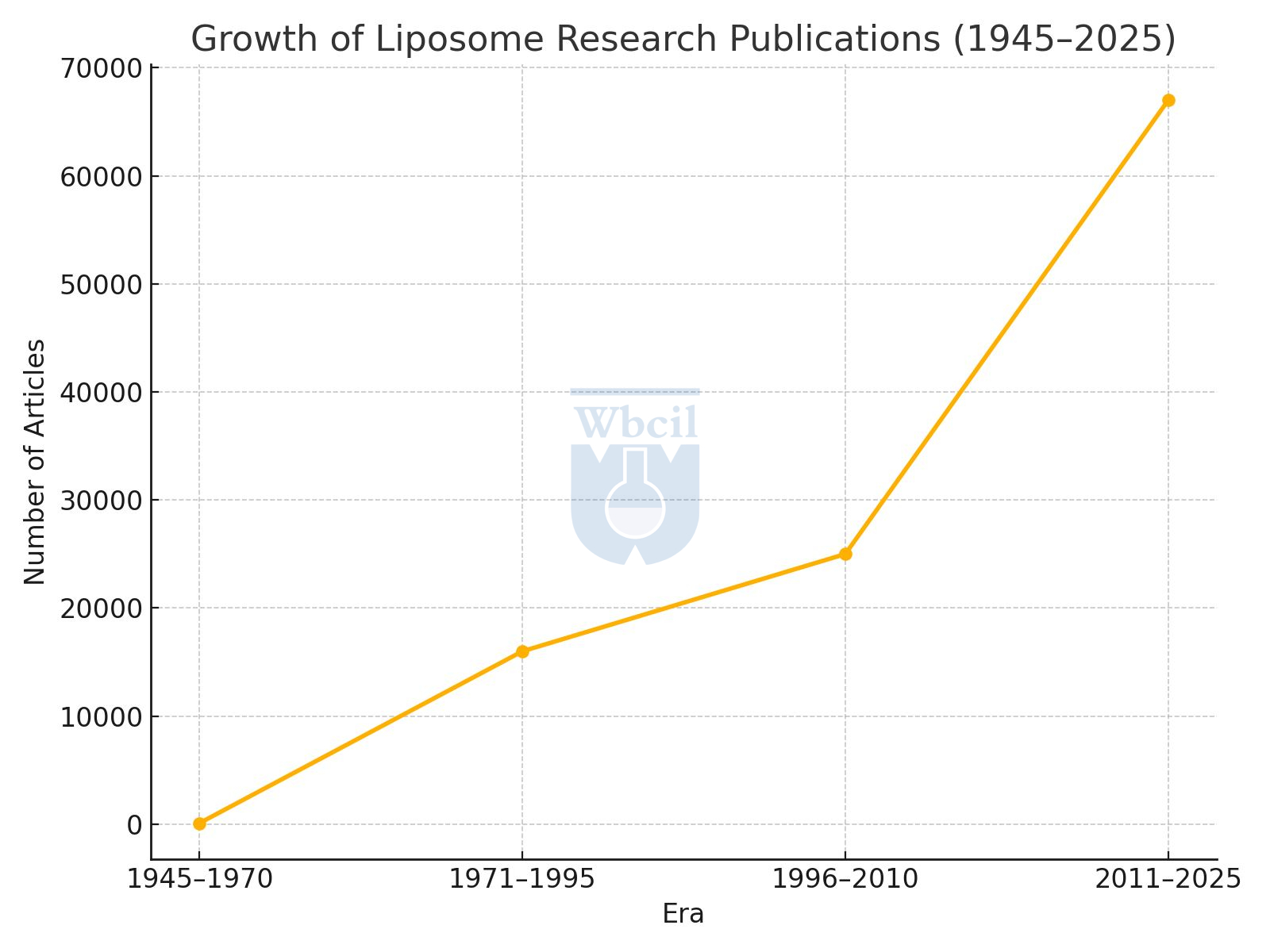
⌛ 1945–1970: The Discovery Era
The journey of liposomes began humbly in the post-war scientific renaissance. Between 1945 and 1970, only about 97 scientific articles were published globally on liposomes. Yet, these early studies laid the essential groundwork, exploring the behavior of phospholipid bilayers and their remarkable ability to form vesicle-like structures resembling biological membranes. This era was purely experimental, focused on understanding the fundamental physicochemical properties of liposomes. It was during this period that the world first realized the potential of these nanostructures as drug carriers — although far from clinical application at this stage.
⌛ 1971–1995: The Exploration Phase
As research intensified, the field transitioned from academic curiosity to applied exploration. Over 16,000 scientific publications emerged globally, marking a dramatic increase in interest. Researchers began to unlock the true potential of liposomes, leading to the first experimental formulations in drug delivery and therapeutics. This era witnessed the introduction of liposomes into early clinical trials, primarily in oncology and infectious diseases. Liposomes were recognized not just as passive carriers, but as versatile and tunable systems capable of enhancing the bioavailability, stability, and therapeutic index of sensitive molecules. This period set the stage for transforming liposomal research into tangible, real-world applications.
⌛ 1996–2010: The Initial Phase of Rising Commercialization
During this critical phase, liposomes shifted from the lab bench to the pharmaceutical market. The world saw the first wave of FDA-approved liposomal drugs, such as liposomal doxorubicin, liposomal amphotericin B and others, which demonstrated improved efficacy and safety over conventional formulations. Their successful commercialization validated decades of research and ignited confidence among scientists and industries alike. The pharmaceutical, nutraceutical, and even cosmetic sectors began exploring liposomal encapsulation to address long-standing challenges like poor bioavailability and drug degradation. This period marked the birth of the liposomal industry as we know it today.
⌛ 2011–2025: The Expansion Age
With the advent of nanomedicine and bioavailability-enhancing technologies, liposomal science entered an era of exponential growth. More than 42,000 scientific articles were published globally in this phase, significantly surpassing all previous records. Innovations in lipid chemistry, PEGylation, targeted delivery, and stimuli-responsive liposomes expanded the scope of applications beyond oncology into vaccines, nutraceuticals, skincare, immunotherapy, and even gene therapy.
The successful deployment of lipid-based systems in mRNA vaccines, most notably during the COVID-19 pandemic, further solidified liposomes’ position as a scalable, safe, and highly effective delivery platform. Today, liposomes are considered indispensable in modern drug delivery, fueling hundreds of commercial products and research pipelines globally.
This remarkable momentum reflects the global scientific community’s confidence in liposomal technology as a cornerstone for future pharmaceutical, nutraceutical, and cosmeceutical innovations.
Global Attention Beyond Academia
In parallel to research, international forums have embraced liposomal innovation, featuring it at prestigious events such as:
- Liposome Research Days (LRD)
- Controlled Release Society (CRS) Annual Meetings
- NanoMed Europe
- SupplySide East / Vitafoods Europe
These gatherings unite scientists, formulators, and business leaders — discussing the latest applications of liposomal APIs in immune health, beauty, cognitive function, and chronic disease management.
The dynamic field of liposomal technology has seen significant advancements in recent years, with global conferences serving as pivotal platforms for knowledge exchange and innovation. At West Bengal Chemical Industries Limited (WBCIL), our Research and Development team actively engages with these international events to stay at the forefront of liposomal research, integrating cutting-edge insights into our product development processes.
Key Global Events Influencing WBCIL’s R&D
- 18th Liposome Research Days 2024
- Date & Location: June 26–29, 2024, at the Technology and Innovation Centre, University of Strathclyde, Glasgow, Scotland.
- Overview: This conference, themed “Delivering Innovation and Driving Applications,” brought together international experts to discuss advancements in liposome-based drug delivery systems. Topics included vaccines, gene therapy, gene editing, chemotherapy, and lipid and membrane biophysics.
- Impact on WBCIL: Insights from this event have been instrumental in refining our liposomal formulations, particularly in enhancing the stability and targeting efficiency of our drug delivery systems.
- 26th International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Date & Location: Scheduled for September 8–9, 2025, in Frankfurt, Germany.
- Overview: This conference focuses on innovative drug delivery systems, including liposomal drug delivery. Discussions revolve around the design, development, and application of liposomes in therapeutic delivery.
- Anticipated Impact on WBCIL: Knowledge from this conference is expected to provide valuable insights into the latest trends and technologies in liposomal drug delivery, guiding our future research endeavors.
- 4th Annual Extra-Hepatic Lipid-Based Nanoparticles Delivery Summit
- Date & Location: June 24–26, 2025, in Boston, MA. Overview: This summit focuses on the development of lipid-based nanoparticles for delivering nucleic acids and gene-editing payloads to extra-hepatic targets. It addresses challenges and innovations in lipid nanoparticle delivery beyond RNA applications.
- Relevance to WBCIL: The summit’s focus aligns with our interest in expanding the applications of lipid-based delivery systems, potentially leading to the development of new liposomal products targeting specific tissues.
What Is WBCIL Working On in Liposomal Innovation?
At West Bengal Chemical Industries Limited (WBCIL), we are proud to contribute to this growing body of science and industry demand. Backed by WHO-GMP and ISO certifications, our research and production teams are leading the way in high-performance liposomal APIs and skincare solutions.
WBCIL’s Liposomal Product Portfolio Includes:
- Liposomal Iron – For anemia and energy metabolism
- Liposomal Calcium – For bone health and cellular function
- Liposomal Magnesium – For stress, nerve health, and sleep
- Liposomal Zinc – For immune support and skin integrity
- Liposomal Glutathione – For detoxification and skin brightening
- Liposomal Vitamin C – For immunity and collagen synthesis
- Liposomal Alpha Lipoic Acid – For antioxidant support and metabolic health
- Liposomal Sunscreen – For advanced skin barrier protection
- Liposomal Serum – For deep-penetrating anti-aging skincare
- And many more under development…
With a strong focus on R&D, WBCIL is also collaborating with global partners to develop custom liposomal formulations tailored to B2B client needs across nutraceuticals, pharmaceuticals, and cosmeceuticals.
How Global Liposomal Events Drive Our R&D Forward?
Participation in major international forums like Liposome Research Days (LRD), Controlled Release Society (CRS) Annual Meetings, NanoMed Europe, and SupplySide East plays a pivotal role in strengthening our in-house innovation.
These events allow our R&D team to:
Stay Ahead of Scientific Trends – We gain access to breakthrough studies, clinical results, and formulation strategies being adopted globally.
Benchmark Our Technology – By analyzing how global leaders are applying lipid-based delivery systems, we ensure WBCIL formulations are at the cutting edge.
Network with Experts – These platforms foster collaborations with academic researchers, pharma formulators, and biotech pioneers.
Incorporate Real-World Insights – Trends in liposomal skincare, RNA delivery, personalized medicine, and chronic disease support help us anticipate market needs.
The insights gathered from these events directly feed into our formulation strategy, clinical research planning, and product pipeline, empowering us to design solutions that are not only effective but also scientifically validated and future-ready.
Conclusion
Staying abreast of international advancements in liposomal technology through active participation in global conferences empowers WBCIL’s R&D team to innovate and enhance our product offerings continually. By integrating global insights, we reinforce our commitment to delivering cutting-edge, effective, and safe liposomal products to meet the evolving needs of healthcare and consumer markets.
